June 26, 2017
The original wearable was an electric torpedo fish.
Yep, that's right. In 46 AD, the personal physician to the Roman Emperor Claudius applied an electric torpedo fish to the Emperor’s forehead to successfully relieve his migraines. Don't believe me? Check out “Thousands of Years Before Modern Electricity, Ancient Romans Used Electrotherapy to Effectively Treat Neurological Conditions”. Anyways, this first “wearable” was actually alive, and the “user experience” was provided by the twitching underbelly of the fish, which was enraged from being pulled out of the sea and which covered the top half of the patient’s face. It filled the patient’s nose with its fishy scent, and delivered at first a painful and then numbing electric shock (roughly the same voltage as a hairdryer dropped in a bathtub).
“These are still the early days of wearable neurostimulation,” an ancient tech blogger would have reported, “But on the bright side, the device, while possessing eyes and teeth, is at least conveniently wireless.”
Heh.
Today we know that electrically stimulating the trigeminal nerve, which runs, in part, beneath the forehead, does in fact effectively suppress migraines. Indeed, the FDA approved the first wearable neurostimulation device to treat migraines in 2012 and like the torpedo fish, the wearable’s electrode is placed on the forehead.
But the idea of using electrical stimulation to affect not only pain perception, but mood and sleep, arose more recently in human history, and in this article, written by technology journalist Kelly Roman, you'll not only discover the fascinating history of electrical stimulation for addressing cognitive issues, but you'll also discover how a small company called “Fisher Wallace” developed a deep brain stimulation device that causes your body to experience a massive natural release of dopamine, serotonin and fall into a state of deep relaxation, no drugs required.
One Flew Over The Cuckoo's Nest
Electroconvulsive therapy (also known as “ECT”) came into use in 1938. The original ECT user experience was significantly worse than having an electric fish placed on your head (as Jack Nicholson vividly demonstrated in the 1975 film One Who Flew Over the Cuckoo’s Nest). That film would effectively brand ECT, and neurostimulation in general, for the next 30+ years as something barbaric, despite the fact that anesthesia is widely used for ECT procedures. Since the 1970’s, in fact, ECT patients have quietly slept through their shock therapy and have woken up feeling better – but you wouldn’t know that from seeing the movie.
The 1970’s also saw the rise of psychiatric medications, which provided a far superior user experience to ECT, at least initially. This innovation helped inspire a small group of electrical engineers to partner with medical researchers to develop more discrete, non-invasive forms of neurostimulation that delivered small, comfortable doses of electricity on a daily basis, as opposed to the infrequent, seizure-inducing doses of ECT.
The idea, in essence, was to achieve a sort of “user experience parity” between a daily antidepressant pill and a daily low dose of electricity. By the mid 1970’s, a number of well controlled published studies began to validate small dose, alternating current electrotherapy, for the treatment of depression and anxiety as well as the treatment of insomnia. A tiny handful of devices, such as the Neurotone 101, emerged from these studies as something that doctors were willing to prescribe, and in 1976, the FDA grandfathered cranial electrotherapy stimulation devices into the modern era of federal regulation.
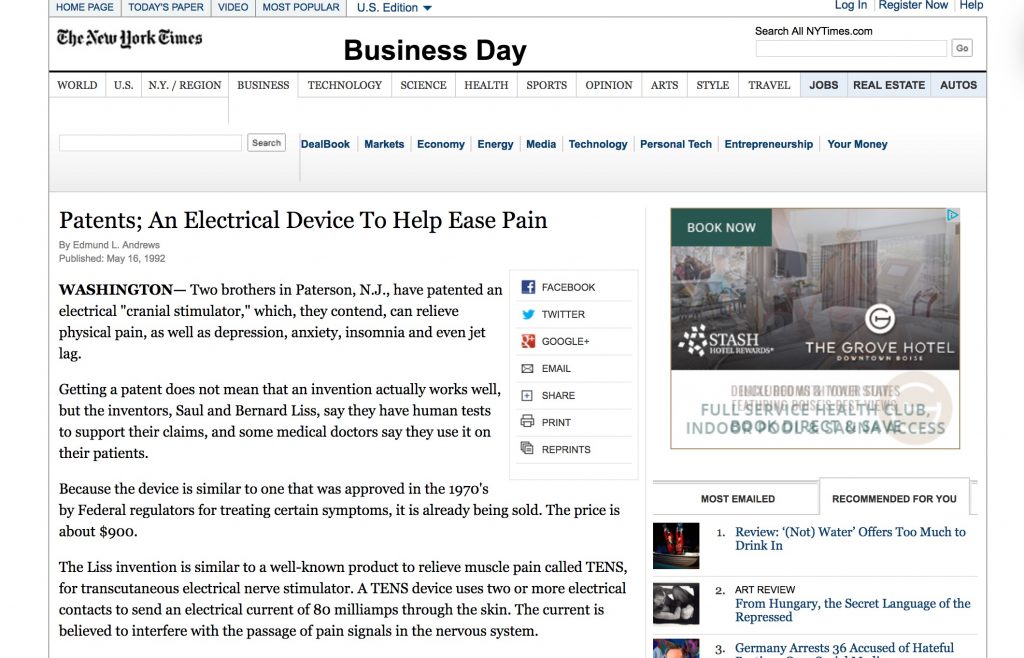
But these early devices were often the size of small typewriters and not well suited for home use, and none were successfully commercialized for the mass market. However, by the 1980’s, a couple of electrical engineers who happened to be brothers, Saul and Bernard Liss, developed a small, portable neurostimulation device that deployed two simple electrodes worn beneath a headband (here's an old-school New York Times article about it). Not only was the device easy and comfortable to use, but it employed multiple frequencies that allowed its alternating current to reach deep within the brain.
The Liss brothers published compelling research demonstrating that their device stimulated the brain to durably produce serotonin and melatonin (required for healthy mood and sleep) while lowering cortisol (the stress hormone). Unlike antidepressant medication, which increased serotonin by inhibiting neuronal receptors from absorbing it, their device caused no serious side effects. They called their device the “Liss Cranial Stimulator®” and in 1990 the FDA cleared it for the treatment of depression, anxiety and insomnia.
Rise Of The Machines: Wearables vs. Drugs
Many people fear using antidepressants not only because of potential side effects, but because of potentially losing their creative edge and their ability to focus. Not only do most patients using devices like the the Liss and other forms of cranial stimulation report significant increases in creativity, but published research proves that alternating current stimulation significantly improves focus and concentration.
Regardless, the initial competition for the Liss Cranial Stimulator® was quite fierce, to say the least. Eli Lilly had just launched Prozac in 1986, the first blockbuster antidepressant. Prozac and the myriad drugs that followed – Paxil, Lexapro, Zoloft, Ambien, Wellbutrin, Xanax and many others – sucked all the oxygen out of the psychiatric marketplace through the use of ubiquitous advertising and physician sales forces. There was no internet to speak of yet, and so launching a product head-on against Big Pharma in the 1990’s was an extremely difficult and extremely expensive task. While technologically stunning, the Liss Cranial Stimulator® remained an obscure, esoteric product.
My business partner Chip Fisher (whose father founded the very well known Fisher Radio company) discovered the Liss Cranial Stimulator® in 2006 and bought the patents from the estate of the Liss brothers, who had since died. Chip and his original partner, Martin Wallace, who was an addiction-ologist, renamed the device the “Fisher Wallace Stimulator®”. Martin Wallace then died a year later from sudden cancer, and I replaced him as co-founder. Chip and I were the only ones left standing, but the product and the company had, in essence, five co-founders.
I had personally come from the world of social media marketing, and had also worked at the scientific journal Nature. The opportunity was clear to me: Facebook and Google allowed us to compete head on with Pharma in a way that was previously impossible. Given the Liss device’s strong scientific data (click here for the impressive research Dropbox) and lack of side effects, I felt like we had a near moral obligation to educate the public about our technology.
Electric cars have been around since the mid 1850’s but it took Tesla, founded in 2003, to redefine the electric car user experience and marketing approach (ie, build a supercar first and iterate down the price point chain) and revolutionize battery manufacturing at scale. So Chip and I drew inspiration from Musk and today still see Fisher Wallace as the early-days Tesla of psychiatric medicine.
In 2009, a handful of prominent psychiatrists in New York City finally started prescribing our device. The highest volume prescriber was Dr. Richard Brown, who served on the faculty at Columbia University School of Physicians and Surgeon, where he still teaches pharmacotherapy. We asked Dr. Brown if he would volunteer an hour of his time to speak to a room full of psychiatrists who had never prescribed our device but were interested in hearing about his experience. Dr. Brown generously agreed, and we hosted the event at Chip’s apartment on the Upper East Side. About 15 psychiatrists showed up to the first talk.
Two months later we hosted another talk, this time with one or two new doctors who had started prescribing the device and agreed to share their experience to a room of 25 new doctors. Soon thereafter, we assembled a medical advisory board for Fisher Wallace Labs that included the Chairman Emeritus of NYU’s Department of Psychiatry, Dr. Robert Cancro.
We also helped fund a 392-subject study at Phoenix House, the largest non-profit drug rehab clinic in the country (see Scientific American's article “Return of Electro-Cures: Symptom of Psychiatry's Crisis?“). The study examined if our device could increase patient retention. The group of newly admitted opioid addiction patients who used our device remained in rehab after 90-days at a rate 50% higher than those who did not use our device.
In the world of addiction rehab, a 50% increase in 90-day retention is a staggering success.
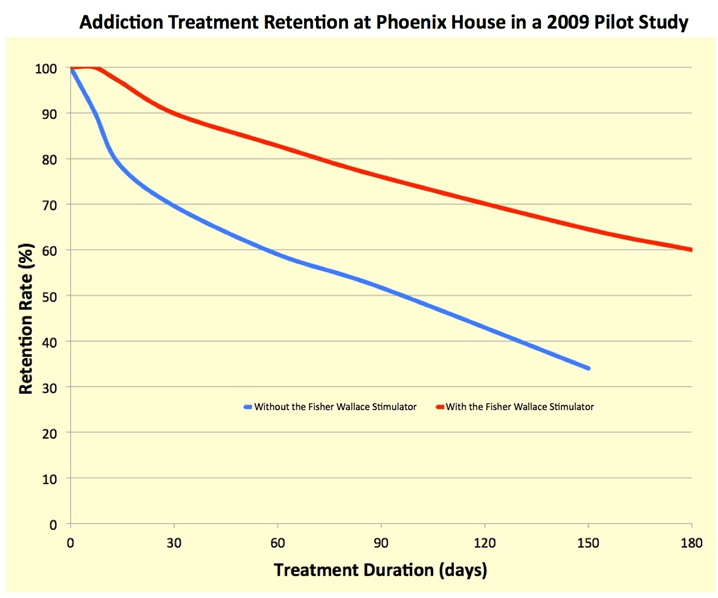
That 2009 study, combined with the biomarker studies published in the 1990’s that proved the device stimulated the brain to produce serotonin and melatonin while decreasing cortisol, positioned the Fisher Wallace Stimulator® as something with both significant clinical and scientific validation.
Electrostimulation Enters The Mainstream
Fitbit was also beginning to find its legs in 2009, and their ability to change consumer perception around what a wearable device actually is, unintentionally helped us position our medical device as a form of wearable technology. That shift in the way we could frame our device allowed us to move beyond the public memory of Jack Nicholson’s performance in “One Who Flew Over the Cuckoo’s Nest”.
In other words, our device was no longer being compared to electroshock therapy, it was now being compared to Fitbit – instead of just tracking your sleep, our device could actually treat insomnia. The influence of fitness trackers on how the public and the media began to think about neurostimulation cannot be understated.
With our new scientific data, doctor and patient testimonials, and wearable vernacular in hand, I began to use Facebook and Google advertising to drive awareness for our technology. The more customers we have, the better “lookalike audiences” we are able to create on Facebook.
By 2012, we had over 2000 prescribers. That same year, a study was published that demonstrated a new mechanism of action for alternating current: stimulation like ours was found to effectively dampen the brain’s default mode network, the center region of the brain that is hyperactive during periods of stress.
In 2015, a landmark study at Mount Sinai Beth Israel Hospital used the device to successfully treat bipolar depression. The study was published in the Journal of Nervous and Mental Disease and literally overnight positioned the Fisher Wallace Stimulator® as a novel, low risk treatment option for 6 million bipolar patients (remember: antidepressant medication is poorly suited for treating bipolar depression because SSRIs notoriously trigger mania and hypomania).
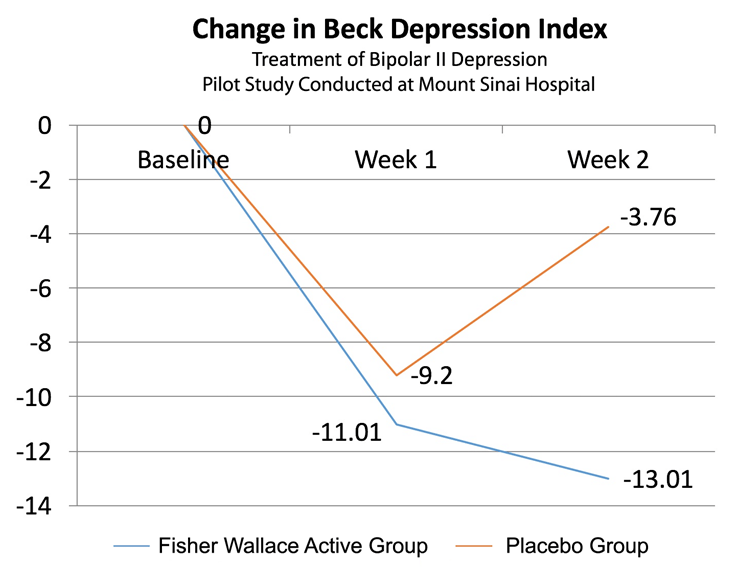
Finally, in 2016, the Fisher Wallace Stimulator® was approved by Medicaid in Maine, the first state-run Medicaid program to approve our device. As a matter of fact, if you live in Maine and are covered by Medicaid, you may obtain a Fisher Wallace Stimulator® at no out-of-pocket expense (visit MaineCare.info).
Today we have 7000 prescribers and 25,000 active patients. Forbes magazine recently named us “one of four technologies innovating mental health” (see “4 Technologies Innovating Mental Health In 2016“). Two of the nation’s leading PTSD experts, Dr. Frank Ochberg and Dr. Stephen Xenakis, have become members of our medical advisory board. We have successfully treated hundreds of veterans, including Navy SEALs and hard-charging, high-achievers like Ben Greenfield – who occasionally need to settle down stress and simply take a nap or fall asleep faster.
I recently began noticing that a few wearable neurostimulation devices were being released by VC-backed start-ups that targeted the general wellness market by avoiding any medical claims. By not making medical claims, these products, such as Thync and Halo (read Ben Greenfield's review here), avoid being regulated by the FDA and may be obtained without a prescription. The downside, in my opinion, is that these devices have very slim clinical track records and very little device-specific scientific evidence. They tend to reference a lot of research that was not performed with their devices in order to make broad effectiveness and safety generalizations, when in fact we know very little about how their specific devices impact human health.
But I understand why start-ups want to avoid FDA regulation and prescription requirements – being regulated by the FDA is often a burdensome, expensive and time consuming process. You need hard science to get FDA-Clearance, and the prescription process makes buying a device more complicated.
At the same time, I believe there is much more value created for the patient by medical device companies like Fisher Wallace that take the time to conduct research and submit to strict FDA regulation.
So the question for me became…
…how can Fisher Wallace offer the accessibility of a general wellness device with the scientific validation of a medical device?
Enter The Circadia – The 21st Century Electric Torpedo Fish
The answer was a device now called the “CIRCADIA®”.
The CIRCADIA®, which Ben Greenfield has now been personally using and trialing for the past 2 months before finally encouraging me to write this article, uses exactly the same technology – the same PCB, the same dosage, the same electrode placement – as the Fisher Wallace Stimulator®, and thus inherits all the scientific and clinical validation of the technology itself.
But CIRCADIA® targets a different population: people who are otherwise healthy but looking to better manage stress and sleep. As a result of its non-medical intended use, CIRCADIA® is not classified as a medical device like the Fisher Wallace Stimulator®, and can be obtained instantly without a prescription (I'll tell you how in a moment) for the following issues:
– Becoming easily agitated, frustrated and moody
– Feeling overwhelmed, like you are losing control or need to take control
– Having difficulty relaxing and quieting your mind
– Feeling bad about yourself (low self-esteem), lonely, worthless and depressed
– Avoiding others
– Low energy
– Headaches
– Upset stomach
– Aches, pains, and tense muscles
– Chest pain and rapid heartbeat
– Insomnia
– Frequent colds and infections
– Loss of sexual desire and/or ability
– Nervousness and shaking, ringing in the ear
– Cold or sweaty hands and feet
– Excess sweating
– Dry mouth and difficulty swallowing
– Clenched jaw and grinding teeth
Better yet, with the CIRCADIA®, we are now able to enter the same general wellness market as Thync and Halo, but with a product that, specifically for stress and sleep, is far superior in terms of clinical and scientific validation. As a matter of fact, a similar but not identical model exists with the drug Nexium. The prescription version of Nexium has the same active ingredients as the OTC version of Nexium. The only difference is the indication language – essentially, how each drug is marketed and how the marketing differentiates consumers.
I believe consumers will rightfully prefer general wellness brands such as CIRCADIA® that clone the technology of established medical devices with strong scientific and clinical track records. We know our technology is safe because it’s already been used by tens of thousands of patients. And we know it works because we have the published research to back it up.

You can use CIRCADIA® as often as you would use the Fisher Wallace Stimulator®: once or twice a day for 20 minutes. I personally use it immediately when I wake up in the morning to get me ready for an intense day, and then again at night as I’m winding down to go to sleep. Before I used this technology, I would wake up at 4AM nearly every morning. I am now able to sleep soundly until my alarm goes off at 6:45AM.
Here's a video that shows exactly how the CIRCADIA® works:
So what does the stimulation from the CIRCADIA® feel like?
Most users will not feel the stimulation, while some may feel a slight tingling at the sponge electrode sites. Users typically feel more relaxed after the first 10 minutes of stimulation, as a result of neurotransmitter production.
Less than 1% of CIRCADIA® users may experience a temporary headache, dizziness or skin irritation at the electrode sites. CIRCADIA® may be used safely in conjunction with any medication. Non-invasive brain stimulation devices such as CIRCADIA® should not be used by people who have implanted medical devices, such as vagus nerve Stimulator®s, deep brain Stimulator®s or pacemakers (such devices may be interfered with by external electrical stimulation). We are confident in this safety information because the same applies to the Fisher Wallace Stimulator® which has been on the market for many years.
How To Get Your Own CIRCADIA®
OK, so here's how you get one for yourself, along with a no-questions-asked 30 day refund and return guarantee. Fisher Wallace Labs has partnered with Ben Greenfield Fitness to offer anyone who discovers this article a $100 discount AND free FedEx shipping on any purchase of CIRCADIA®.
Just go to Circadia.info and when you checkout, enter the coupon code GREENFIELD.
We have live customer service via phone, chat and email from 10AM – midnight EST on Mondays-Fridays, and from noon – midnight EST on the weekends. You can also feel free to leave any comments or questions below this article, or to send me questions at [email protected] and I’ll read and respond within 24 hours.
Yours in better sleep and less stress,
Kelly Roman
Co-Founder Fisher Wallace Labs

This blog post was written by Kelly Roman, Co-Founder of Fisher Wallace Labs, an FDA-regulated medical device company based in New York City. Recently cited for producing “one of four technologies innovating mental health” by Forbes magazine, Fisher Wallace Labs has just introduced its first wearable device for stress and sleep management that may be obtained without a prescription, called CIRCADIA®. Customers may receive a $100 discount and free FedEx shipping when they purchase CIRCADIA® by entering the coupon code GREENFIELD at checkout on Circadia.info.


You said in your article above:
How To Get Your Own CIRCADIA®
OK, so here’s how you get one for yourself, along with a no-questions-asked 30 day refund and return guarantee. Fisher Wallace Labs has partnered with Ben Greenfield Fitness to offer anyone who discovers this article a $100 discount AND free FedEx shipping on any purchase of CIRCADIA®.
Just go to Circadia.info and when you checkout, enter the coupon code GREENFIELD.
—————————————————
When I did that, the price of $495 stayed the same–the coupon code did not lower it at all, so I did not order. Why didn’t it work?
Hi Don! They're actually running a special deal now where you get a $300 discount plus free shipping, which is better than what the code would give you. Thanks for the question!
I have the fisher Wallace circadia and I absolutely love it! I’m bipolar and I’m no longer depressed. I’m off my antidepressant now-finally! My mood is stable with just one pill now . I’m sleeping SO much better than I used to and I had awful insomnia that I treated with Xanax, thorazine, and thc edibles before. Thanks to Fisher Wallace all that medicine is in the past.
Fisher Wallace’s refund policy is not nearly as generous as they’d like you to believe. They just raised the restocking fee to 15%, it takes 5 weeks to get a refund, and if you don’t post-mark the device within 2 days of getting your return approved, they will reject it. There are other devices out there at a lower price point that do the same thing.
What are your thoughts on the Alpha Stim versus Fisher Wallace?
Fisher Wallace is the only one I've used and recommend.
Hi Ben, great info. I’m really curious if the circadia helped get you more deep sleep. I’ve been tracking my sleep with ŌURA ring, and struggle to get more than a few minutes of deep sleep most nights.. did you see any direct impact on deep sleep cycles ? Thanks!!
Absolutely. They also have great customer support and a no-questions-asked 30 day refund and return guarantee, so I'd definitely give it a try.
How much more deep sleep did you get Ben?
It jacks it up about 2 to 3% but more importantly it's amazing for naps.
Also the return policy is not 100% refund. There are restocking fees and non refundable costs shown on the website. FYI
Ben,
You mentioned that your HRV score was 104 after using the Fisher Wallace — what was the app or program that you’re using to track the HRV?
Thank you,
Adam
https://greenfieldfitnesssystems.com/?s=naturebea…
Great, thanks.
Do you have a recommendation for the least damaging chest strap to use with the app? (I.e. no/low bluetooth energy, etc..)?
Thank you,
Adam
https://www.amazon.com/gp/product/B007S088F4/ref=…
Cochrane carried out a review in 2014 and found “no high quality clinical trials comparing CES with sham CES in people with acute depression. Currently, there is insufficient evidence to support the use of CES in treatment of acute depression.”
Lots of negative reviews: https://www.drugs.com/answers/anyone-fisher-walla…
The Cochrane review is not peer reviewed (it was not published by a reputable scientific journal), and employed article exclusion criteria specifically designed to exclude ALL studies from consideration, including double blind, placebo controlled clinical trials – by excluding all studies, it concludes there are none that show evidence of effectiveness; there is no medical definition of “acute depression”, as opposed to actual diagnosed depression disorders such as major depressive disorder and bipolar disorder – “acute depression” is a created term to service their desire to exclude studies. Essentially, the Cochrane review is well known as a smear campaign against wearable neurostimulation, funded and published by political opponents to the CES industry. Cochrane cofounder Peter Gotzche vehemently opposes all forms of psychiatric intervention, and had publicly stated that Eli Lilly has “murdered more than 200,000 people” (you can find the video on YouTube)
I’d recommend that you read peer-reviewed literature such as the 2015 bipolar study by Galynker and the 2014 depression and anxiety study by Barclay (unless, of course, you are purposely trying to advance the Cochrane article)
Here are two peer reviewed studies on the NIH database:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4892…
https://www.ncbi.nlm.nih.gov/pubmed/24856571